TEKLYNX tracking software
{{product.productLabel}} {{product.model}}
{{#if product.featureValues}}{{product.productPrice.formattedPrice}} {{#if product.productPrice.priceType === "PRICE_RANGE" }} - {{product.productPrice.formattedPriceMax}} {{/if}}
{{#each product.specData:i}}
{{name}}: {{value}}
{{#i!=(product.specData.length-1)}}
{{/end}}
{{/each}}
{{{product.idpText}}}
{{product.productLabel}} {{product.model}}
{{#if product.featureValues}}{{product.productPrice.formattedPrice}} {{#if product.productPrice.priceType === "PRICE_RANGE" }} - {{product.productPrice.formattedPriceMax}} {{/if}}
{{#each product.specData:i}}
{{name}}: {{value}}
{{#i!=(product.specData.length-1)}}
{{/end}}
{{/each}}
{{{product.idpText}}}
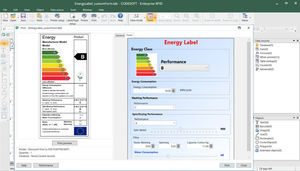
... printers for use with CODESOFT barcode label software thanks to TEKLYNX' continual printer driver development with all major printer manufacturers. Enterprise integration Integrate CODESOFT barcode label software ...
TEKLYNX

From asset tracking to inventory control, the possibilities are endless with BACKTRACK Check-in check-out software gives you the power to track the exact location, movement, and usage of any item. ...
TEKLYNX

... quality and transparency of medical devices in the healthcare supply chain through label design, label approval, and label tracking standardization. As a result, all medical device labels, with the exception of custom-made ...
TEKLYNX

... serialization and traceability for DSCSA was November 27, 2023. The FDA announced a one-year delay in enforcing the electronic tracking requirement under the DSCSA. The enforcement of an interoperable package-level electronic ...
TEKLYNX

... /Lot Number Pack/Harvest Date GS1-128 barcode in human- and machine-readable Supplier ID # and Tracking #
TEKLYNX

Easily create and print GS1-compliant barcode labels TEKLYNX label design software for GS1 Barcodes Create GS1-128, GS1 DataBar, GS1 DataMatrix barcodes, UPC barcodes, QR codes, and more Built-in ...
TEKLYNX

... stations in order to print agreed upon labels AIAG outlines the symbology recommended for automotive part identification and tracking. This standard defines the minimum requirements for marking or labeling individual ...
TEKLYNX
Your suggestions for improvement:
Receive updates on this section every two weeks.
Please refer to our Privacy Policy for details on how DirectIndustry processes your personal data.
- Brand list
- Manufacturer account
- Buyer account
- Our services
- Newsletter subscription
- About VirtualExpo Group



Please specify:
Help us improve:
remaining